Stained Fixed Cells Then Fix Again
Gear up Now? Fix Later? - Considerations for the Use of Paraformaldehyde Fixation in Flow Cytometry
- Staining intracellular targets (e.one thousand. − intracellular cytokine staining, phosphorylation targets) - the cells demand to be stock-still prior to the permeabilization of the cells.
- Convenience − for scheduling purposes, you need to fix the samples in the centre of an experiment so you tin can go along to clarify them later.
- Disinfection − when you lot're working with infectious samples (i.east. HIV-infected patient samples), it's a routine procedure to pre-fix the sample prior to analysis for sanitation purposes.
- Timepoint − when y'all're looking at a progressive biological process over a time-form (e.thou.- phosphorylation of Protein A, X hours after stimulation Y), you'll want to "freeze" all processes using fixation so you get a more than authentic snapshot of what'southward going on at different timepoints.
- It's Saturday night - your buddy Chad is having a birthday fustigate at the Legendary bar downtown starting at 8pm. It'south eight:30 already, you're in the lab, and take just finished harvesting your samples. I mean… yous have no choice, right?
-
- What concentration of fixative should I use for my experiment?
- When in your staining procedure should you lot set your sample in the duration of your protocol?
- Ultimately, how can these decisions potentially affect my catamenia staining?
- What concentration of fixative should I use for my experiment?
In this blog, we'll explore these questions related to fixation, and perhaps some nutrient for thought for your next period cytometry experiment when yous run into a fixation dilemma.
A solution ranging from 1-4% PFA is typically used for fixation of samples for flow cytometry. In the instance of sanitizing infectious samples, concentrations equally low as 0.37% can effectively disinfect samples from HIV-infected patients ane . Incubation for up to 45-60 minutes with one% PFA, and 15-20 minutes with four% PFA (e.grand. BioLegend'due south buffer) is sufficient to fully prepare the cells, and the cells tin either exist used for downstream processing (permeabilization for intracellular targets) or stored for time to come analysis at this phase.
During a routine flow cytometry staining procedure that involves surface marking staining, there are several steps where y'all tin make up one's mind to set your samples - prepare after surface marker staining, or fix before .
In some instances, the decision for when you lot tin set is... stock-still. For instance, if you're analyzing phospho-targets, bounden of certain surface antigens past antibodies can alter intracellular signaling pathways shortly subsequently contact, thus leading to an artifact in your phosphorylation results. In which instance, you lot should fix the cells before the improver of the surface antibodies (which is why our recommended protocol to use for our True-Phos™ Perm Buffer to analyze intracellular phosphorylation targets is designed so that the fixation step precedes other procedures). Each has its own benefits and drawbacks, but here's a couple of things to sentry out for in each example.
Fixation BEFORE staining - epitope alteration:
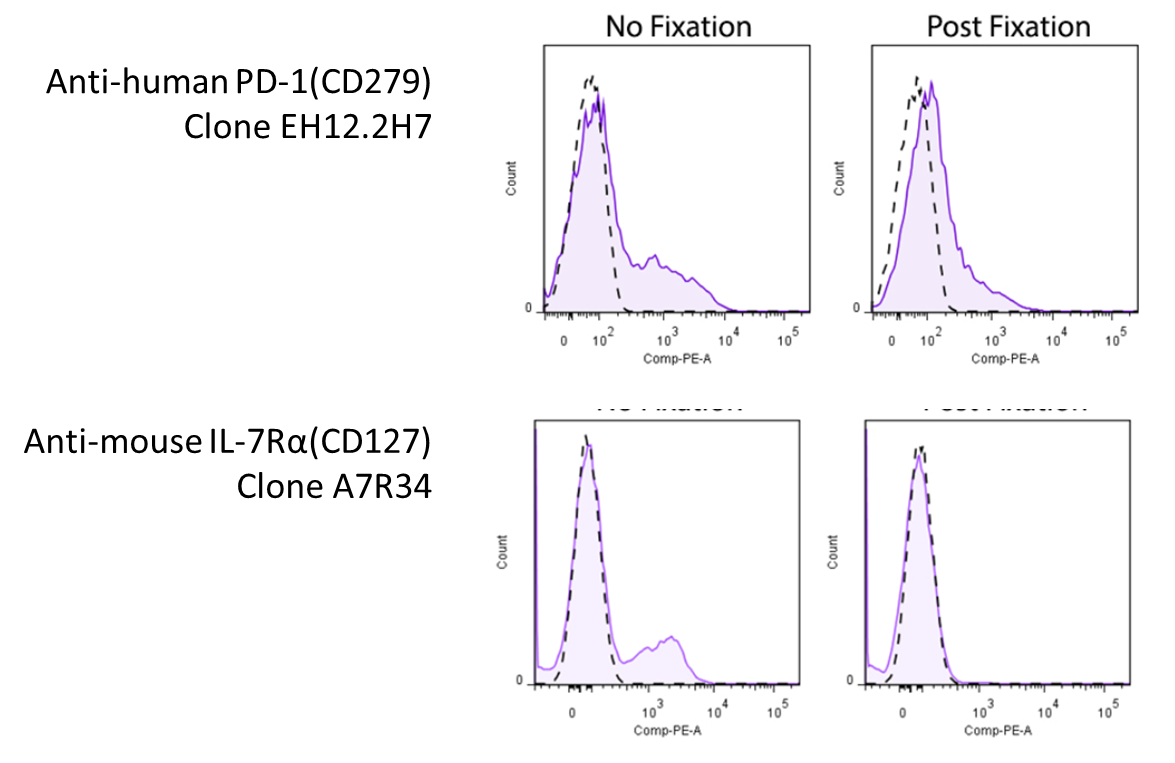
Representative plots for target cells stained with (Postal service-Fixation) or without 4% PFA fixation.
Since this effect depends on the clone of antibody y'all're using, you will want to look effectually to meet whether there's any information pertaining to the antibody you're using. However, yous might be in luck! We've compiled a set of information and information (where available) on whether a particular antibody's binding is affected by 4% PFA fixation, which can be found on our Fixation webpage. For clones not listed on our site, yous may want to consult the literature.
Fixation Subsequently staining - fluorophore stability
To combat the issue of tandem breakup upon fixation, BioLegend has developed the FluoroFix™ Buffer - a PFA-based fixative buffer specially formulated to mitigate signal quenching by sensitive fluorophores.
Do you lot accept any other questions regarding fixation procedures for your flow cytometry work? Any additional tips and considerations to add hither? Let us know at tech@biolegend.com!
mcintirelacir1946.blogspot.com
Source: https://www.biolegend.com/en-us/blog/fix-now-fix-later-considerations-for-the-use-of-paraformaldehyde-fixation-in-flow-cytometry
0 Response to "Stained Fixed Cells Then Fix Again"
Publicar un comentario